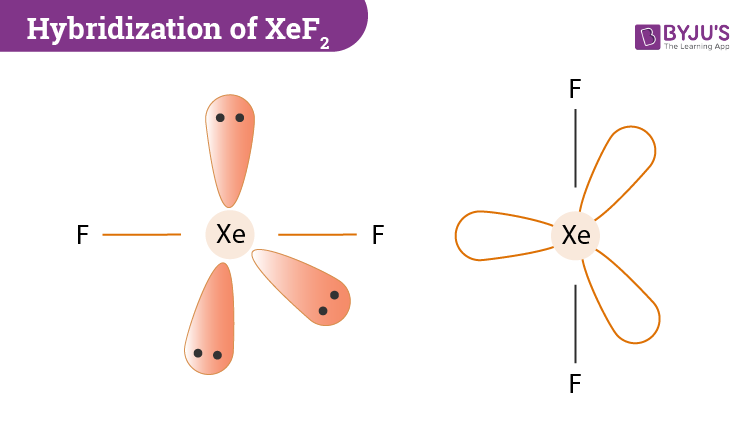
What is the shape of the XeF2 molecule, and the total number of the lone pair present on XE in a XeF2 molecule? - Quora

SOLVED: The XeF2 molecule has a trigonal bipyramidal electron domain geometry and a linear molecular geometry. The FXeF bond angle is molecule is and the

Your Turn! A central atom has two lone pair of electrons around it and two single bonds to other atoms. What is the electron pair geometry around the central. - ppt download

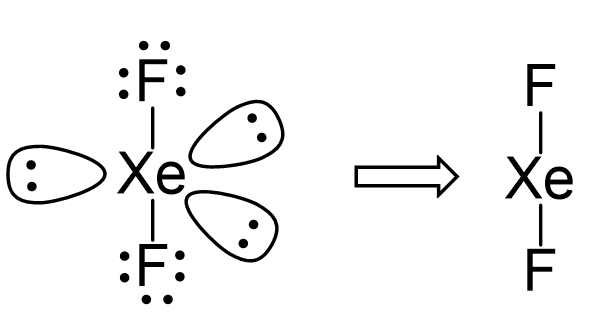
For the Molecule Xef2 : (I) Draw the Structure of the Molecule Indicating the Lone Pairs. (Ii) State the Hybridization of the Central Atom. (Iii) State the Ge.Ometry of the Molecule. -