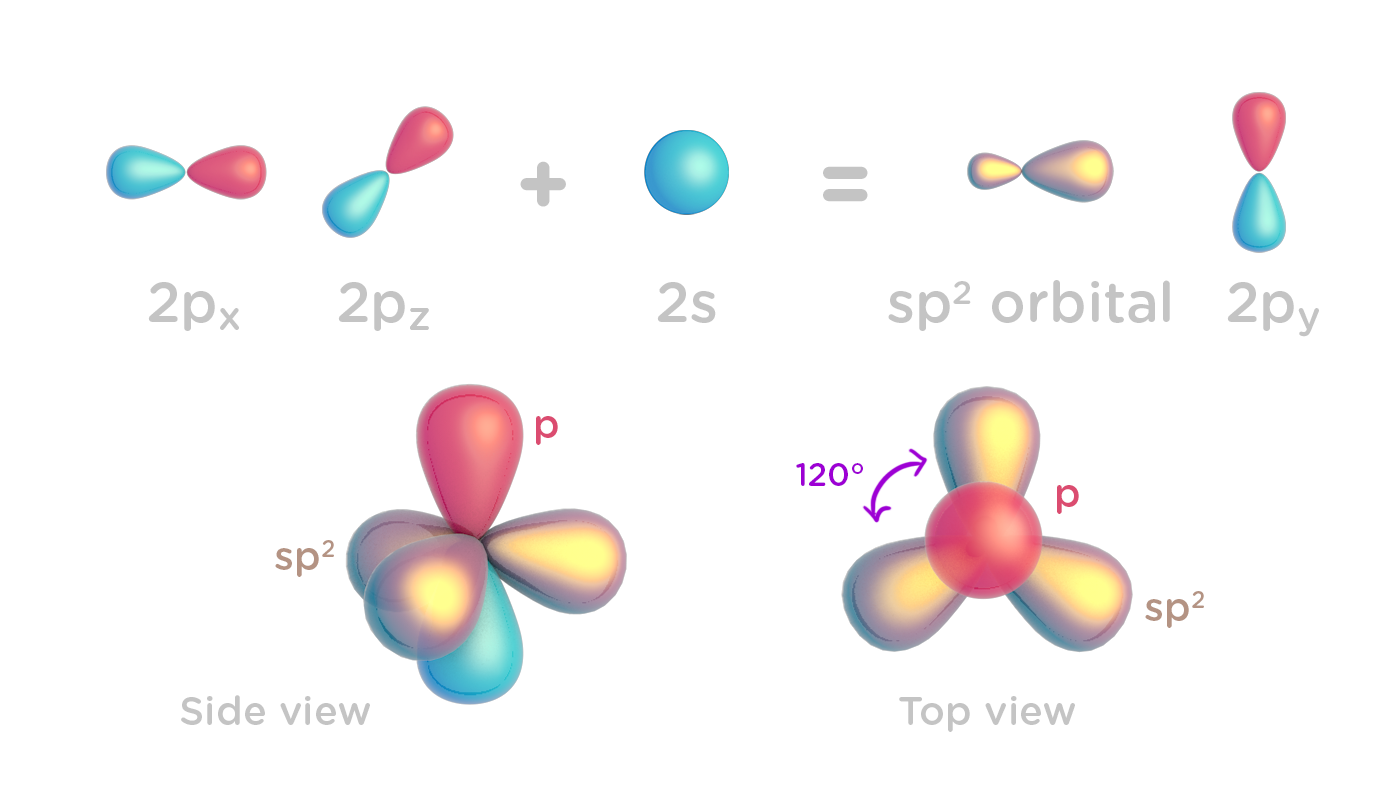
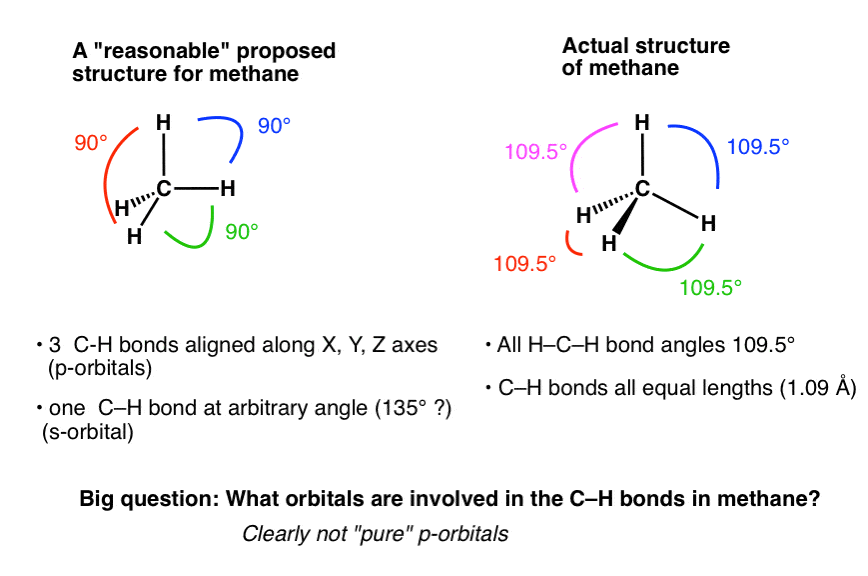
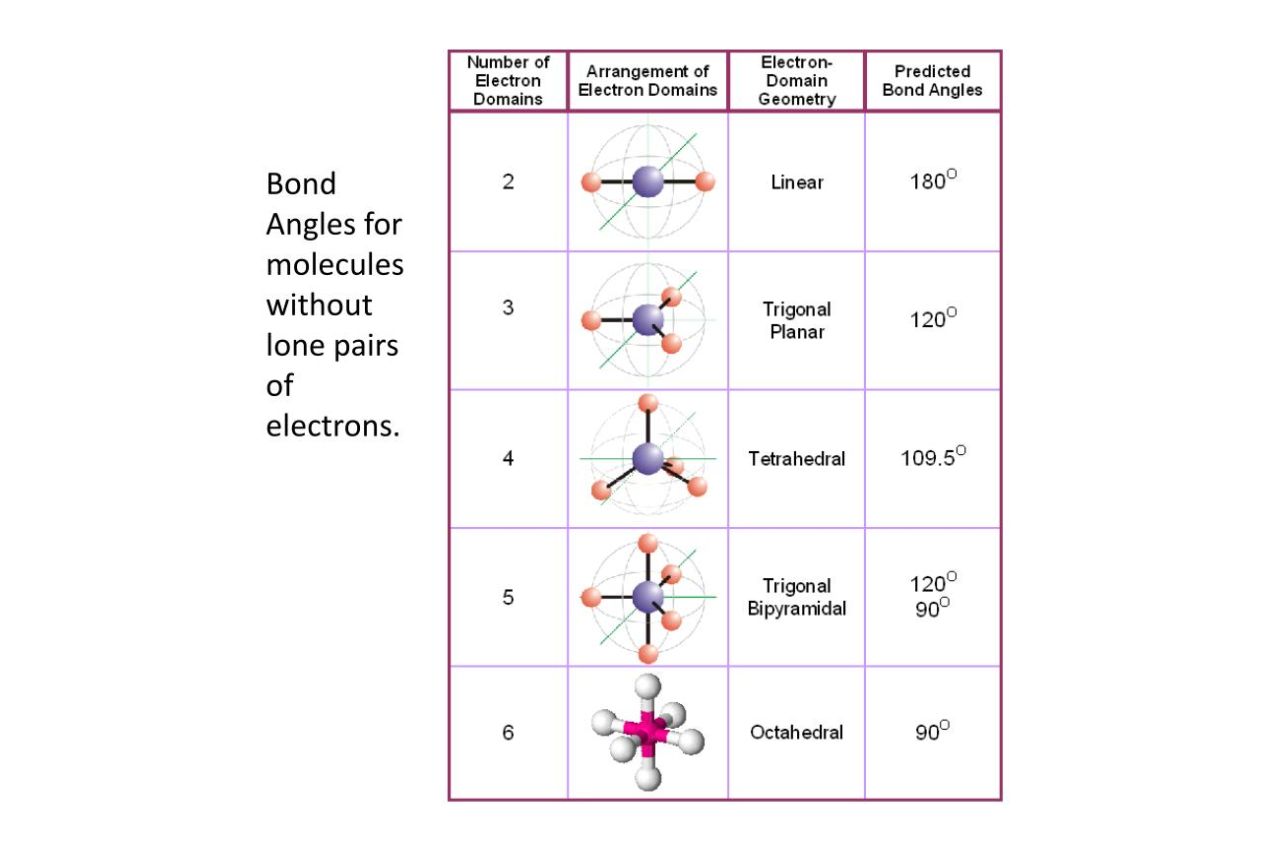
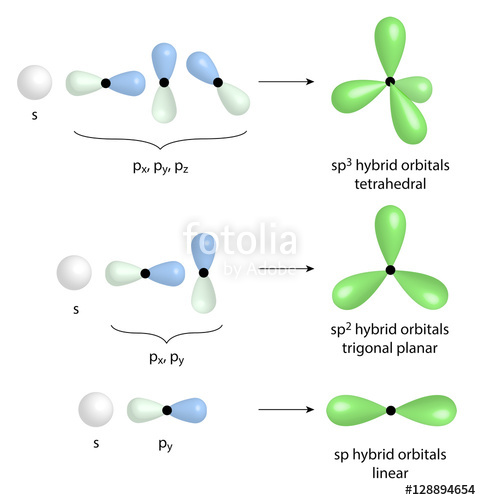
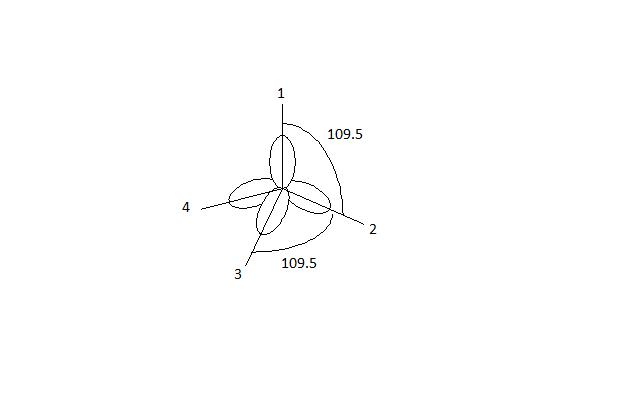
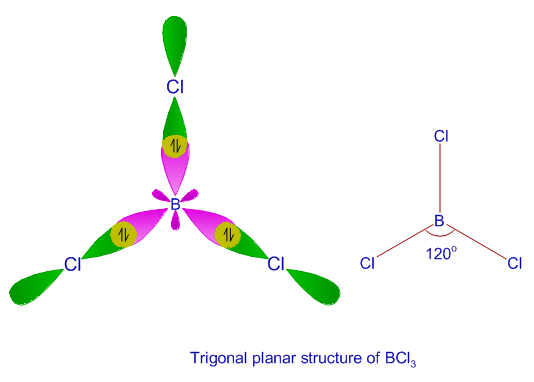
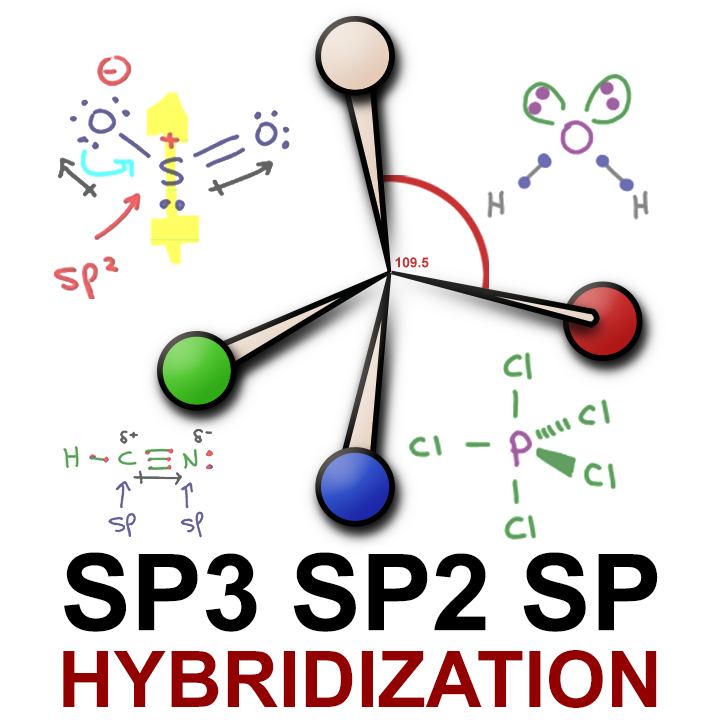
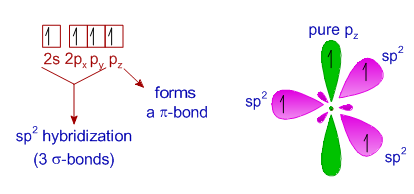
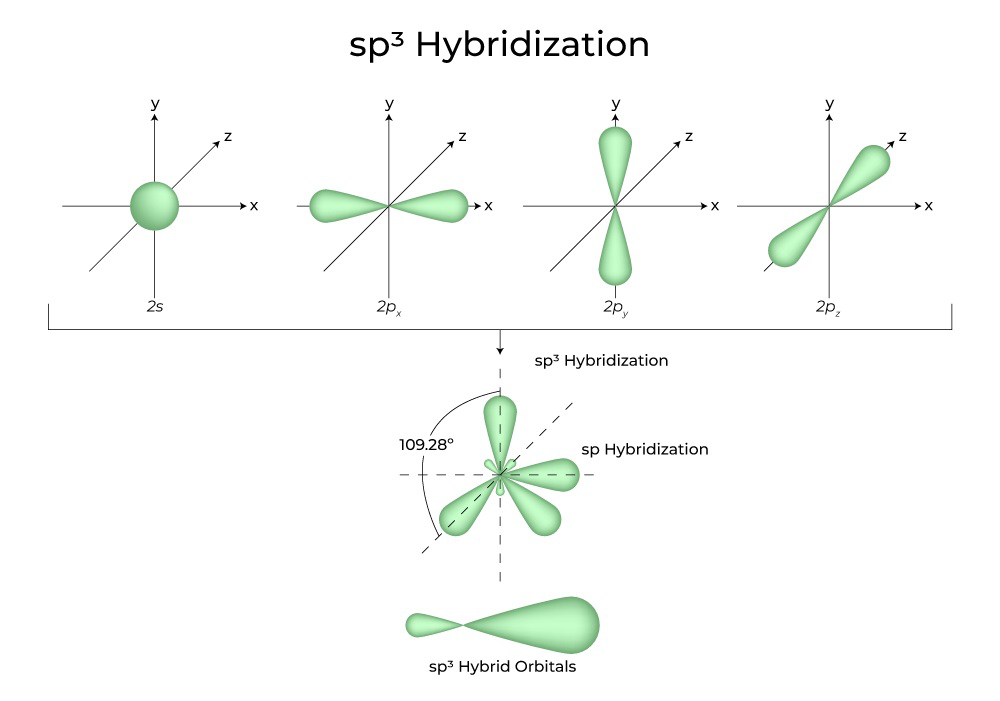
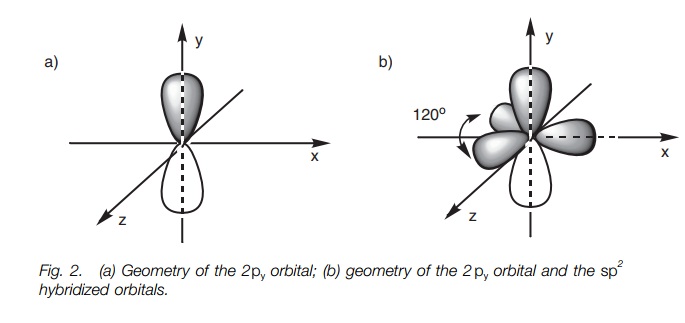
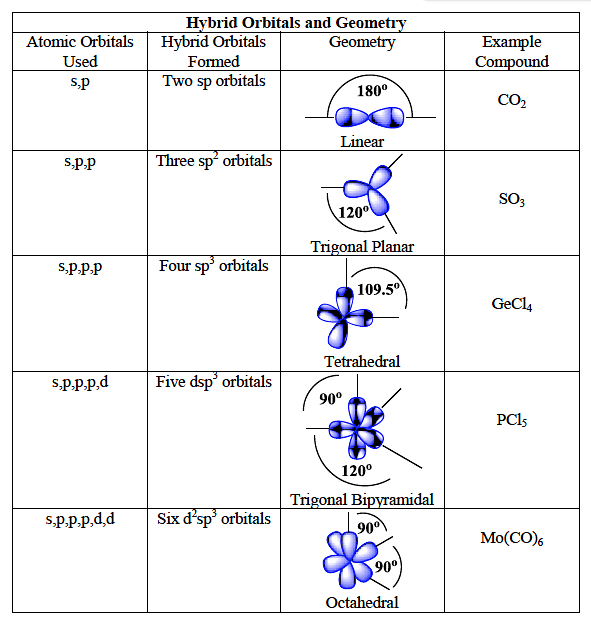
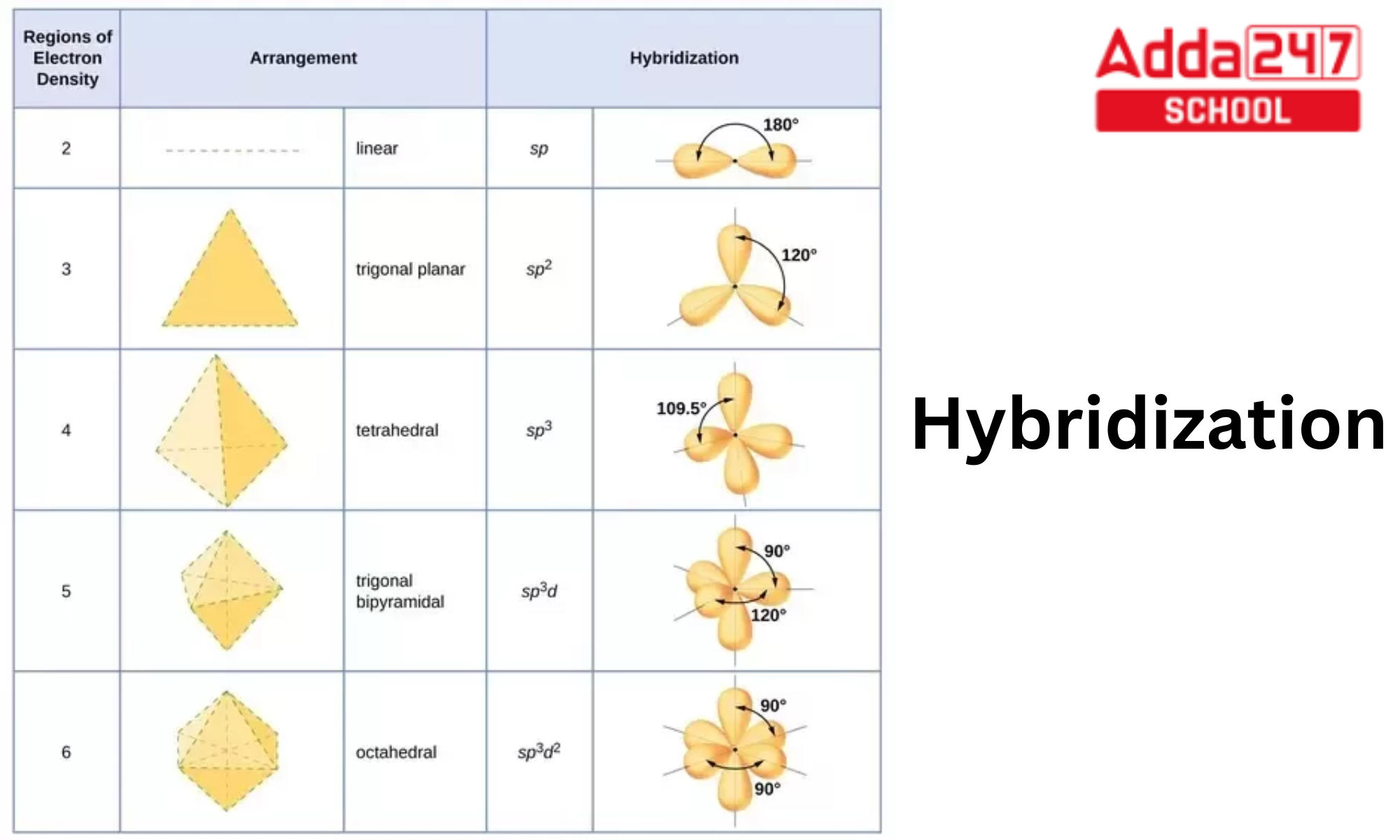
1: Schematic view of the sp 2 hybridization. The orbitals form angles... | Download Scientific Diagram

BF3 - Boron trifluoride hybridization structure shape bond angle adichemistry chemical bonding - YouTube
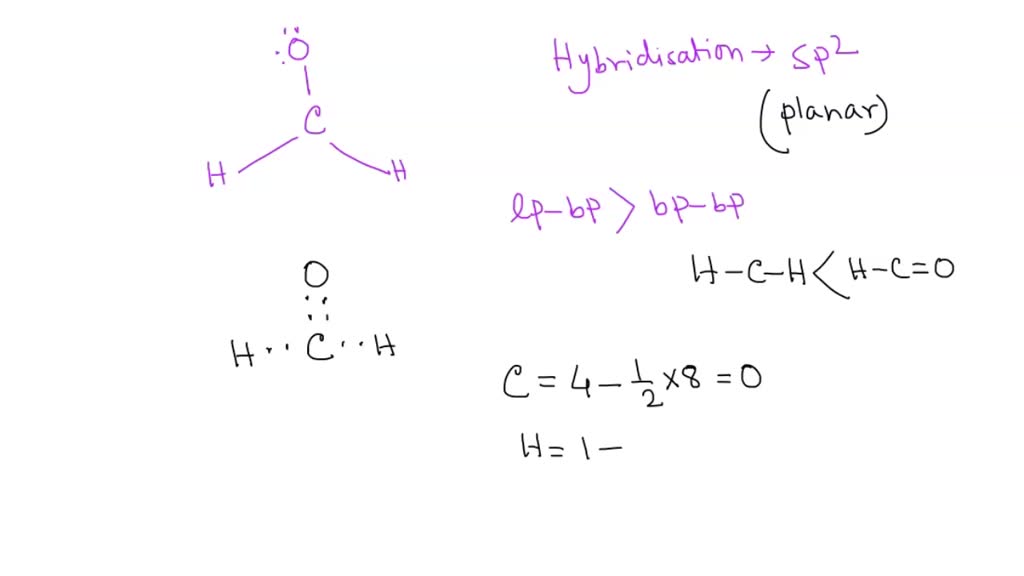
SOLVED: What is true about the CH2O molecule? Carbon utilizes sp2 hybridization. The H-C-H bond angle is expected to be slightly less than the H-C-O bond angle. The molecule contains 3 sigma

a. For the given molecule, what is the hybridization at the atom numbered 1? Enter sp2, sp3, ... b. For atom 1, what Is the bond angle? (degrees) | Homework.Study.com