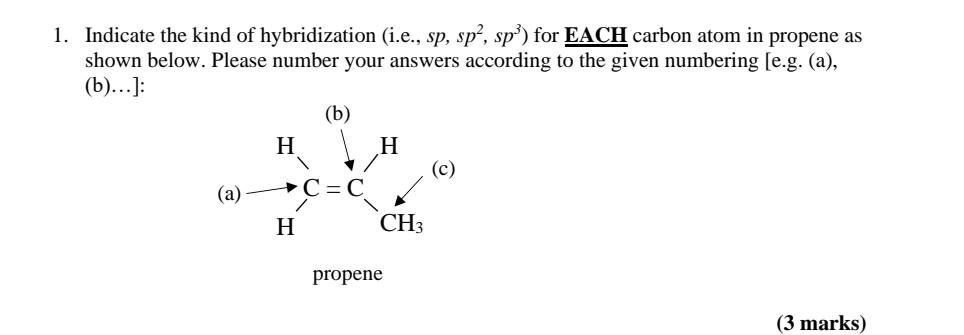
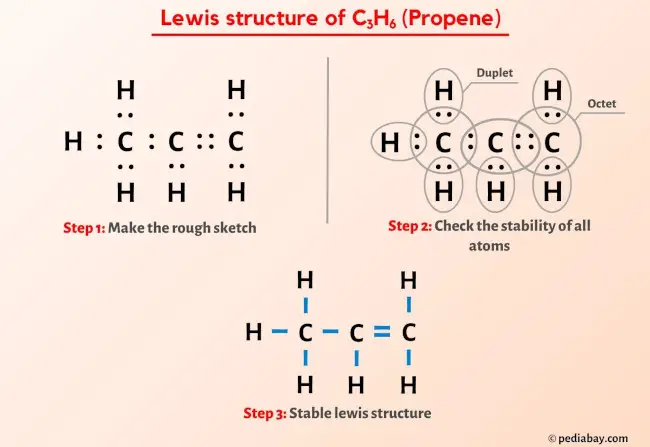
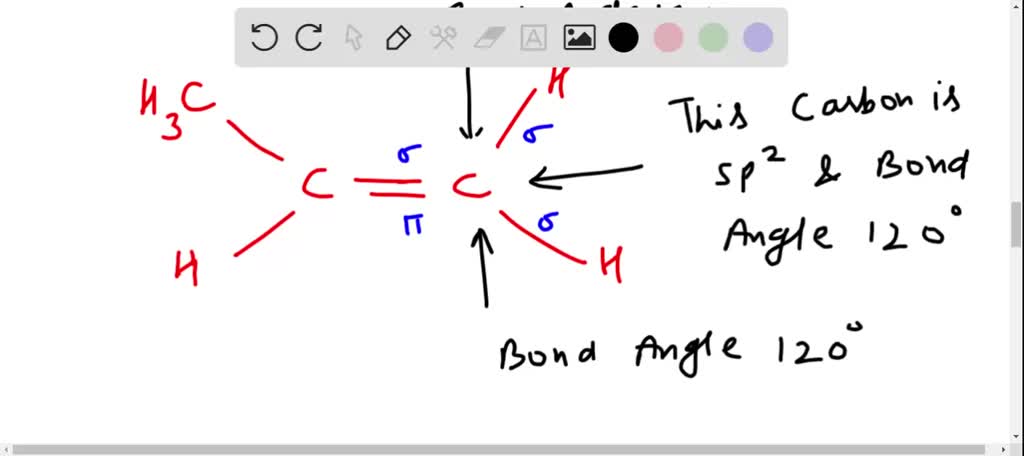
✓ Solved: Draw a line-bond structure for propene, CH3CH=CH2; Indicate the hybridization of the orbitals...
In the displayed formula of propene, why are the two hydrogens on the right, drawn at an angle? Do I need to replicate these angles when draw the displayed formula of propene? -
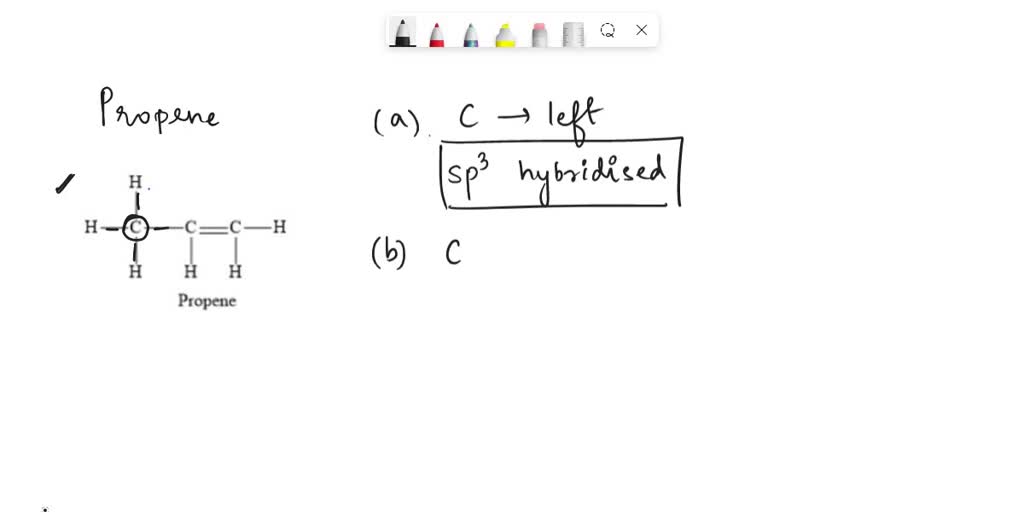
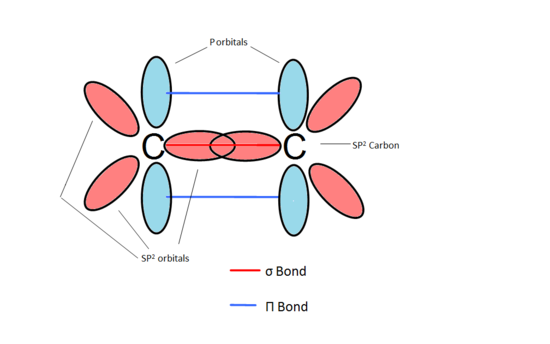
SOLVED: 21 What is the hybridization for each carbon in propene? Carbon on the lefi Carbon in the center Carbon on the right C=C–H H Propene 22 What is the hybridization for
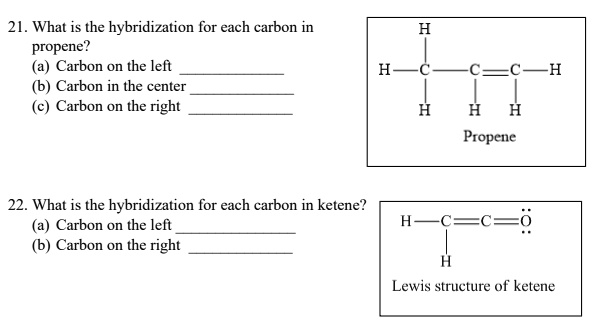
SOLVED: 21 What is the hybridization for each carbon in propene? Carbon on the lefi Carbon in the center Carbon on the right C=C–H H Propene 22 What is the hybridization for

How exactly is this carbon considered sp2 hybridized when it has 3 substituents and a lone pair? : r/OrganicChemistry
✓ Solved: Draw a line-bond structure for propene, CH3CH=CH2; Indicate the hybridization of the orbitals...

Orbital Hybridization Practice Problems - Organic Chemistry #stemeducation #stem #chemistry - YouTube

Draw the molecular shape of propene and determine the hybridization of the carbon atoms. Indicate which orbitals overlap with each other to form the bonds. | Homework.Study.com