The structure of $IBr_{2}^{-}$ involves hybridization of the type:(A) $s{{p}^{3}}$(B) $s{{p}^{3}}d$(C) $s{{p}^{3}}{{d}^{2}}$(D) None of these

Structure of IF_4^+and hybridization of iodine in this structure is, respectively, (A) linear, s ... - YouTube

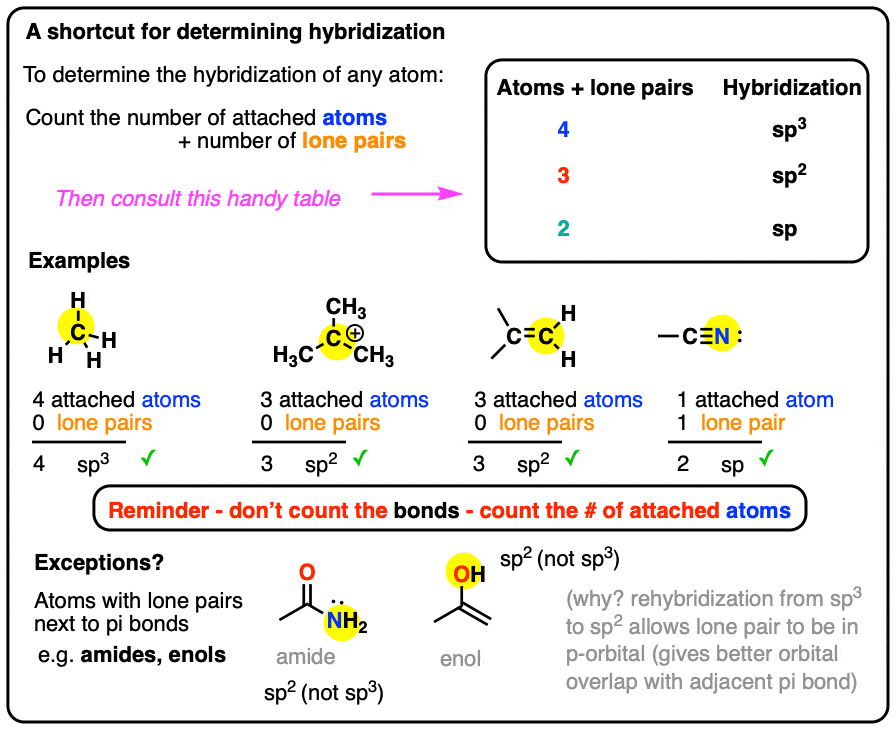
SOLVED: What is the hybridization of the iodine atom in the Lewis structure shown below? To answer the next three questions, refer to the Lewis structure at the top of page 8.

Iodine pentafluoride IF5: Molecular Geometry - Hybridization - Molecular Weight - Molecular Formula - Bond Pairs - Lone Pairs - Lewis structure –