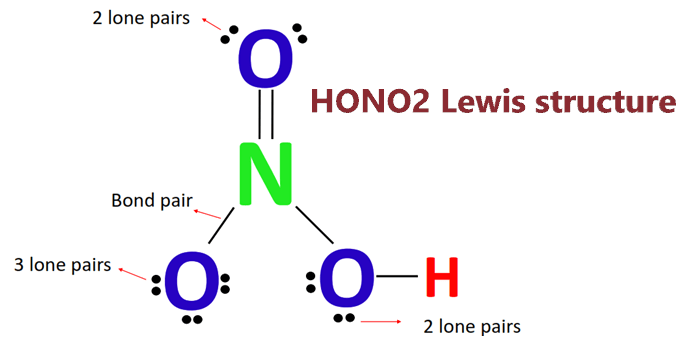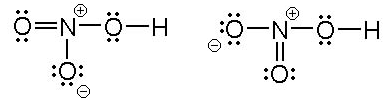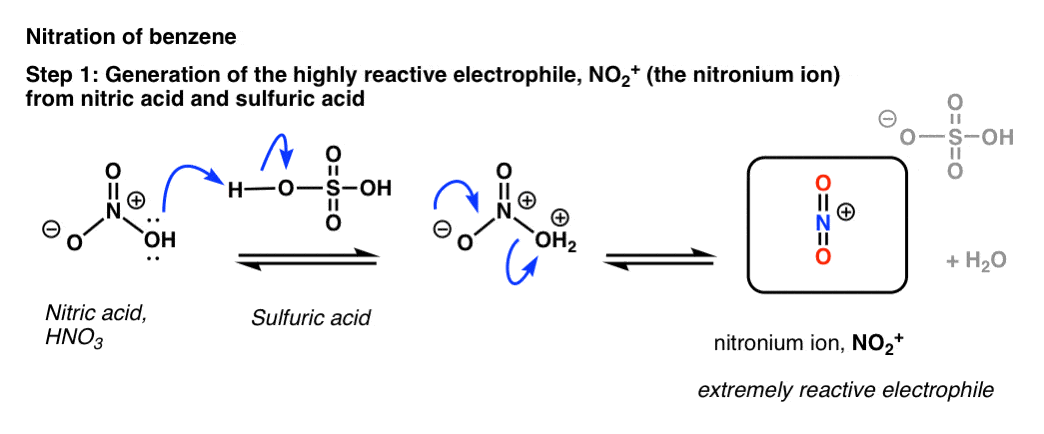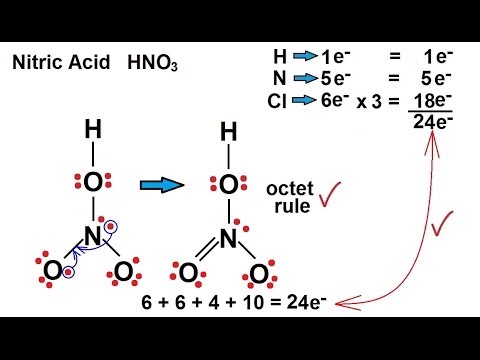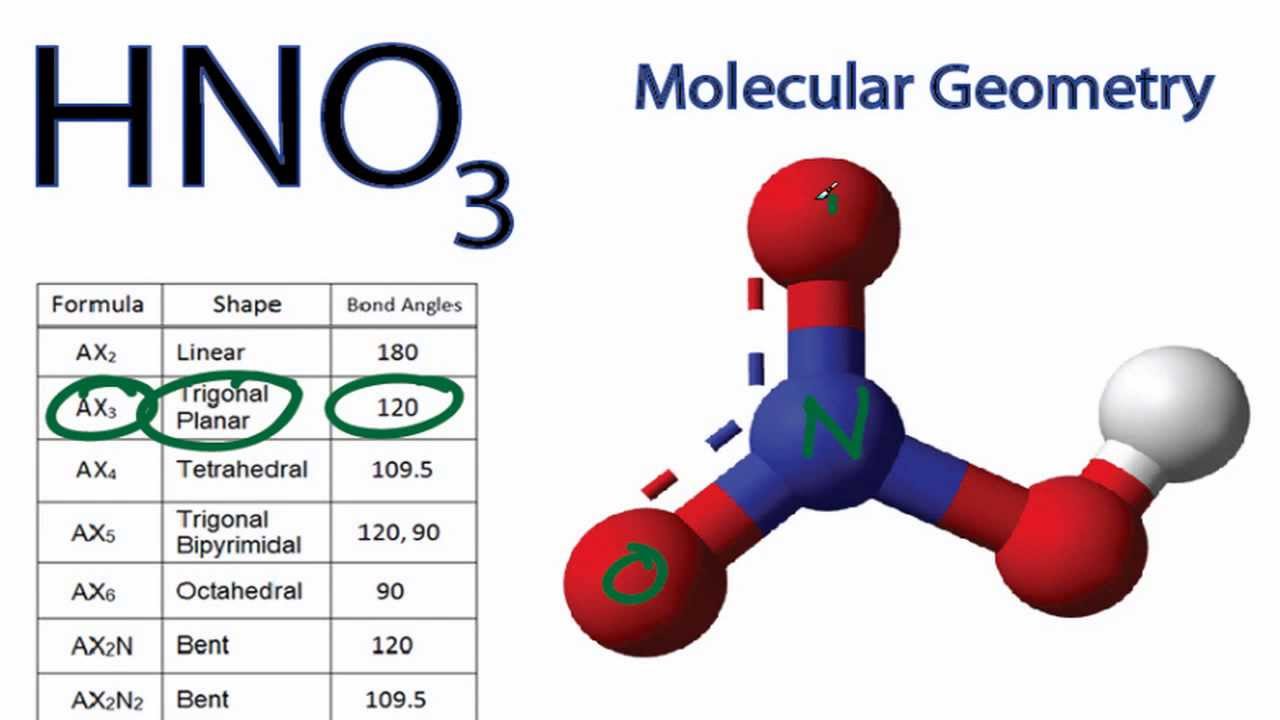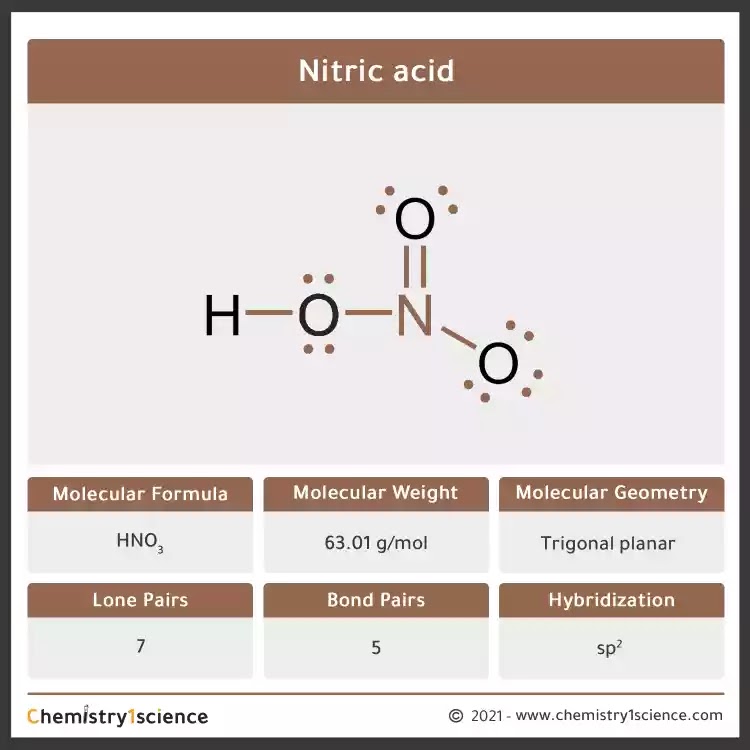
Nitric acid: Molecular Geometry - Hybridization - Molecular Weight - Molecular Formula - Bond Pairs - Lone Pairs - Lewis structure – infographic
In HNO3 why does the nitrogen atom give electrons to the third oxygen when it already satisfied its valency with two oxygens one with a single bond and one with a double

SOLVED: What is the electron-pair geometry about the nitrogen atom in the HNO3 (nitric acid) molecule. Hint: The nitrogen atom is the central atom and the hydrogen atom is attached to an